The African Union has on Monday pressed the European Union (EU) for a review of its Green Pass policy which disqualifies Africans from accessing the continent, based on the type of COVID-19 vaccines received.
The granting of the EU Digital COVID Certificate “Green Pass” applies only to vaccines that have received EU-wide marketing authorisation.
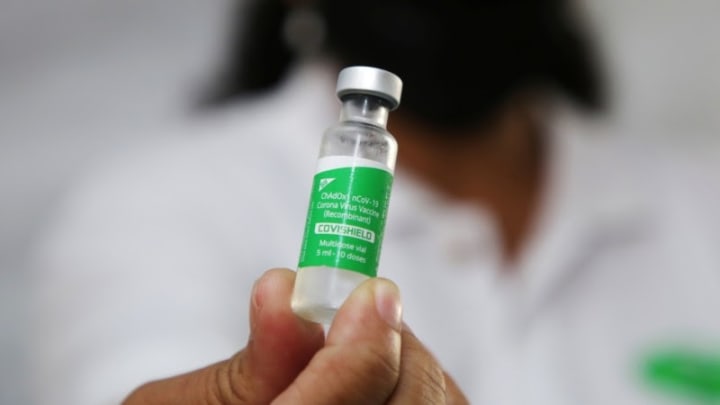
In other words, it excludes the specific kind of AstraZeneca Covid-19 vaccines administered in Africa because it’s produced under the license of the Serum Institute of India, despite being of the same make-up.
These vaccines were largely donated through the global facility for vaccine distribution, COVAX, with support from even from the EU.
Read Also: EU pledge brings fresh vaccine donation to COVAX to 180m
“AstraZeneca vaccine (ChAdOx1_nCoV-19) produced and authorized in Europe (Vaxzervria) is included, the same formation of the vaccine (Covishield) produced under license by the Serum Institute of India (SII), is excluded. Under such regulations, persons who received Covishield, despite being able to demonstrate proof of vaccination, would continue to be subject to public health restrictions, including limitations of movement and testing requirements, with considerable administrative and financial implications,” the African Union Commission and the Africa Centres for Disease Control and Prevention (Africa CDC) said in a joint statement.
The bodies explained the current applicability guidelines put at risk the equitable treatment of persons having received their vaccines in countries profiting from the EU-supported COVAX Facility, including the majority of the African Union (AU) member States.
Given that the Covidshield vaccine has been the backbone of the EU-supported COVAX contributions to the AU member states’ vaccination programmes, AU and ACDC said the development was worrying.
The Serum Institute of India production is designed to serve India and lower-income countries and the SII may not apply for EU-wide market authorisation, meaning that the inequalities in access to “Green Passes” created by this approach would persist indefinitely, the statement read.
The Covidshield vaccine, alongside the versions of the AstraZeneca/Oxford COVID-19 vaccine produced by AstraZeneca-SKBio (Republic of Korea), was one of the first available candidates considered safe and efficacious through the World Health Organisation’s Emergency Use Listing (EUL) process.
Both candidates received approval as early as 15 February 2021 with the explicit goal of rolling it out through the COVAX facility, providing access to lower-income countries and making the global population safe from COVID-19.
The African Union and Africa CDC, therefore, urge the EU Commission to consider increasing mandatory access to those vaccines deemed suitable for global rollout through the EU-supported COVAX Facility